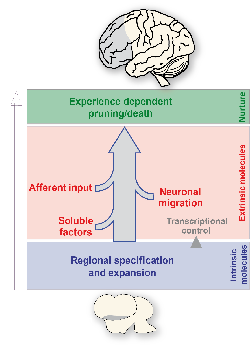
Understanding the processes underlying the development of our complex central nervous system, base of the most sophisticated perceptual and behavioural functions, is a major goal in neurobiology. The generation and maturation of the nervous system involves the negotiation of cells through several consecutive developmental processes: proliferation, migration, differentiation, and formation of proper connections. Brain maturation and arealization is a multistep process that is integrated by intrinsic programs of gene expression, invasion of progenitors generated elsewhere and afferents from other systems guided by extrinsic cues (Fig. 1). Hence, targeted mutation of an intrinsic cue or disruption of the major in- and output systems can produce functional neurological deficits similar to those resulting from brain impairment including epilepsy, autism, AD(H)D, schizophrenia, Parkinson’s or intellectual disability. The identification of the many genes involved in brain arealization now allows for integration of neurodevelopmental defects in mice and humans into a single cellular framework.
Research lines
1) Identify and characterize molecules involved in brain arealization
A) There will be an ongoing search for molecules that are differentially expressed in particular brain areas during development. Focus will be on those brain areas known to be involved in neurodevelopmental disorders (e.g. prefrontal cortex). These areas will be obtained by microdissection.
B) From the obtained RNA sequencing data, a list will be generated of potential interesting intrinsic and extrinsic genes. Expression will be assessed in mouse brains through RT-PCR, in situ hybridization and immunohistochemical combined with morphometric approaches.
2) Unravel the mechanisms of development of specific brain areas
A) First, the expression data described above will hint towards i) what intrinsic factors target certain downstream extrinsic factors (overlapping expression) and ii) what guidance cue receptors bind to certain ligands (complementary or partial overlapping gradient expression). Subsequently, biochemical studies can be undertaken to prove i) transcriptional control of specific guidance cues and ii) a receptor-ligand interaction within a certain area. The latter can be approached by performing immunoprecipitations (to prove interaction), stimulation studies (to show e.g. downstream phosphorylation) and cellular/tissue binding studies (to prove binding in vitro and in vivo).
B) Depending on time and place of expression, proliferation (neurospheres/BrdU) and/or migration assays, can be undertaken to pinpoint further the actual significance of the interaction. In addition, expression of guidance receptors can reveal topographic projections, which can be recapitulated by the insertion of retrograde tracers such as DiI (red) and DiO (green). Three-dimensional co-cultures of explants taken from the origin, guidepost and/or target area of the above mentioned target area will reveal the chemotropic nature of the interaction shown by either attraction or repulsion of the outgrowing neurites. Cues of interest transfected into 293HEK cells can then be used to evoke attraction or repulsion in explant assays.
C) To follow the development of a certain brain area in vivo, cells of this area of interest can be transfected with fluorescent constructs at a certain developmental time point. To this end, we are taking advantage of the novel innovative technique; in utero electroporation (IUE)-mediated gene transfer to monitor proper brain development. After harvesting certain days post-transfection, cellular characteristics such as proliferation, migration and connectivity of transfected cells can be assessed. Combined with a fluorescent Nissl counterstain to demarcate brain regions, cell-type specific markers can be used immunohistochemically to identify areas and cells. Cell division and birthdate (BrdU) as well as cell death (TUNEL) can be examined.
3) Elucidate mechanisms of pathogenetic development of brain areas involved in neurodevelopmental disorders (lines in collaboration)
A) The cerebral cortex in general and the prefrontal cortex (PFC) in specific are involved in the pathology of many neurodevelopmental disorders. The intricate organization of specialized areas in the mature cortex is achieved by coordinate programs in cells that seed this structure during development. However, the molecular basis for this complex developmental script remains unclear. Together with Dr T Kleefstra (Department of Human Genetics) we generated a research line investigating the function of genes implicated in intellectual disability cohort and their role in corticogensis.
B) Cognitive dysfunction and mood disorders in Parkinson’s disease (PD) and Lesh-Nyhan Disease (LND) can be contributed, least in part, to impaired dopaminergic innervation of the PFC. Many therapies, including cell replacement strategies, affect the dopaminergic wiring of the PFC, however little is known about the molecular basis of developing mesocortical projections. Furthermore, recent studies have shown that the habenulae play an important role in cognition. Damage of the habenulae can cause cognitive disturbances similar to those seen in schizophrenia. Through a close collaboration with Dr J. Visser (Department of Neurology) we try to comprehend the forces acting upon PFC and habenulae innervation will work towards a better understanding of mesocortical/habenular-directed cognitive behaviour in order to restore lost functions.
C) The low activity short (s) allelic variant of the serotonin transporter (5-HTT) linked polymorphic region (5-HTTLPR) in humans is associated with reduced 5-HTT expression, structural and functional changes in the PFC and anxiety, and predisposes to these disorders under conditions of environmental adversity. It is widely speculated that these 5-HTTLPR s-allele prefrontal changes are implicated in anxiety due to serotonin-mediated neurodevelopmental changes in the PFC and its circuitries. Together with Dr J Homberg (Department of Cognitive Neuroscience) we are analysing prefrontal circuitry maturation across developmental stages both in vitro as well as in vivo and link this to anxiety in the 5-HTT knockout model by using in utero electroporation.
The experiments outlined are aimed at fully characterizing the molecular forces that act upon the development of the three systems described above thereby moving towards a better understanding of the molecular basis of neurodevelopmental psychiatric disorders.

